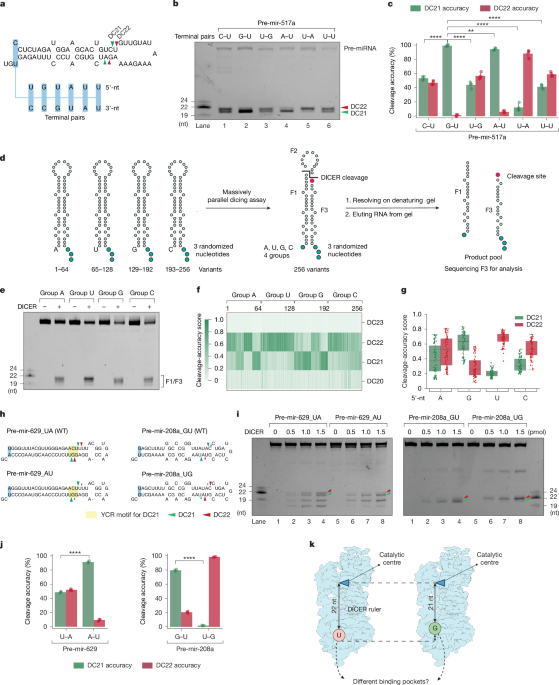
RNA interference (RNAi) depends on DICER, an essential enzyme that processes RNA precursors into small regulatory RNAs. DICER cleaves RNA precursors according to the 5′-end counting rule, in which RNA length is measured from the 5′-end1–3. Previous work proposed a single 5′-end binding pocket that disfavours guanosine (5′-G), leading to cleavage inaccuracies4. Here we show that 5′-G promotes precise cleavage for many substrates. Using massively parallel dicing assays and cryo-electron microscopy, we identify a conserved guanosine-favoured (G-favoured) binding pocket in DICER, distinct from the previously described uridine-favoured (U-favoured) pocket. Together, these pockets influence the alignment between 21-nucleotide and 22-nucleotide cleavage registers, expanding the mechanism of small-RNA biogenesis in metazoan DICERs. We also find that conflicts between 5′-end binding and RNA-motif recognition can trigger RNA conformational adjustments that preserve accurate cleavage-site selection. In addition, conformational adjustments of the double-stranded RNA-binding domain (dsRBD) and PAZ domain help to align substrates with the catalytic centres for precise double-strand cleavage. These results show that the DICER cleavage mechanism integrates dual 5′-end binding pockets, RNA-motif influence and domain motions, advancing our understanding of microRNA biogenesis. Cryo-electron microscopy and massively parallel assays shed light on the mechanism by which DICER, a key enzyme in the RNase III family, cleaves RNA at precise locations to produce small RNAs. Plasmid construction The pXG-10×His-DICER plasmid was generated by inserting a DNA sequence encoding human DICER (amino acids 25–1922) and a sequence encoding a 10-histidine tag at the N terminus of DICER into the pXG plasmid using the In-Fusion clon... [22011 chars]