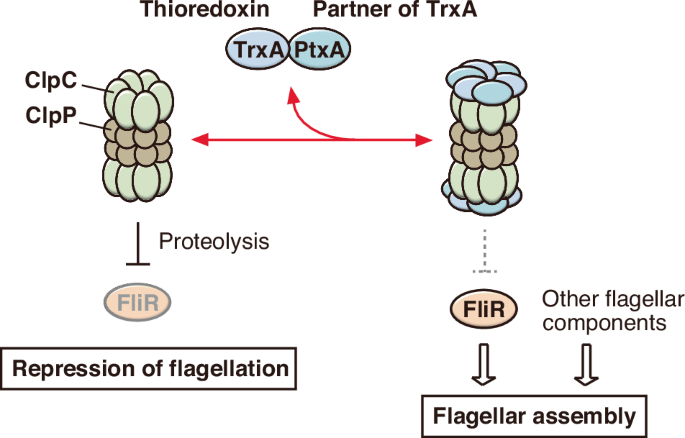
The filamentous bacterium Actinoplanes missouriensis produces flagellated spores, which become zoospores after release, in sporangia. However, the regulatory mechanism of spore flagellation remains unclear. Here, we report that a thioredoxin, TrxA, and its partner protein, PtxA, are crucial for flagellation. Most spores of trxA and ptxA null mutants are non-flagellated, despite normal transcription of flagellar genes. Gene complementation tests show that the EKVEQ motif in TrxA, but not thioredoxin activity, is indispensable for flagellation. Analysis of motile suppressor strains obtained from both mutants reveals that ClpC, a chaperone component of the Clp protease complex, and FliR, a flagellar secretion gate component, are involved in the regulation of flagellation. TrxA and PtxA interact with each other and bind to ClpC in bacterial two-hybrid analysis. Based on the experimental results, we propose a working model for flagellar assembly control by TrxA and PtxA in A. missouriensis. The actinomycete Actinoplanes missouriensis produces flagellated spores that become zoospores after being released from sporangia. A genetic study has revealed that a thioredoxin and its partner protein control flagellar assembly. Silverman, M. & Simon, M. I. Bacterial flagella. Annu. Rev. Microbiol. 31, 397–419 (1977). Chevance, F. F. & Hughes, K. T. Coordinating assembly of a bacterial macromolecular machine. Nat. Rev. Microbiol. 6, 455–465 (2008). Macnab, R. M. How bacteria... [11852 chars]