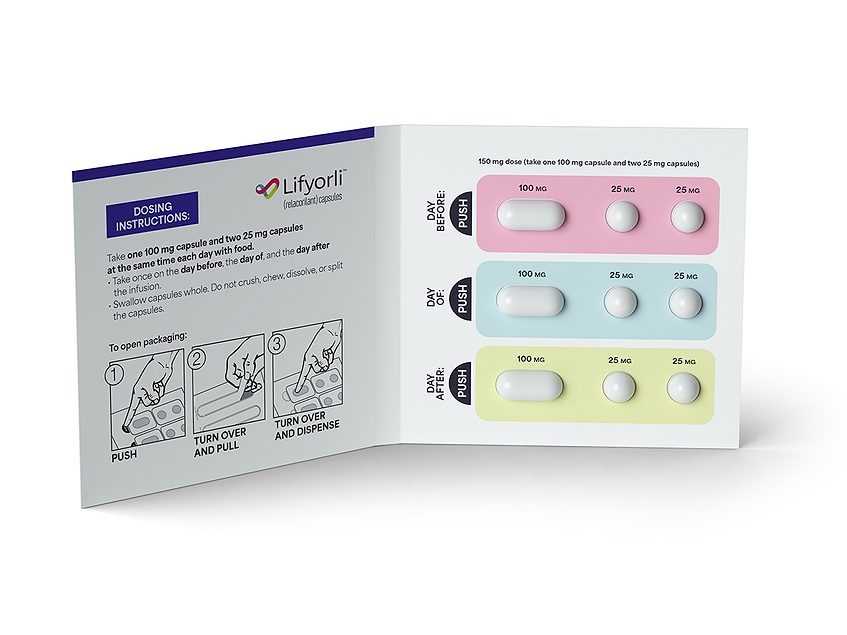
The U.S. FDA approved Corcept Therapeutics Inc.’s oral, selective glucocorticoid receptor antagonist relacorilant nearly four months ahead of schedule for adults with platinum-resistant epithelial ovarian, fallopian tube or primary peritoneal cancer.