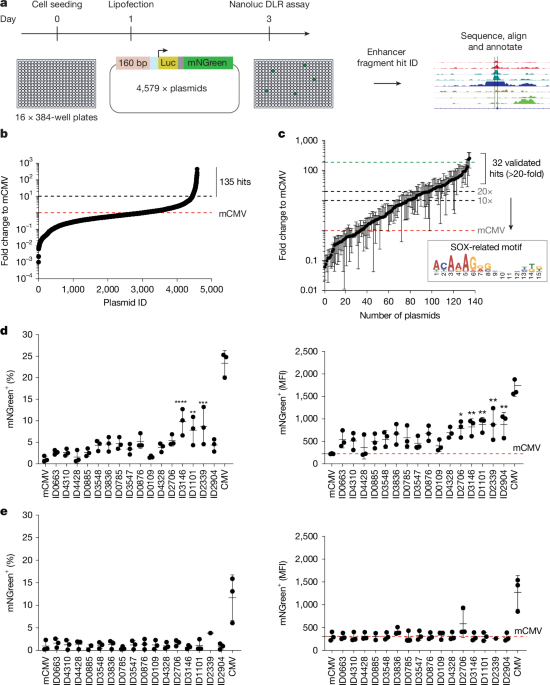
Cell-type-specific promoters are used in gene therapy to restrict expression of the therapeutic payload. However, these promoters often have suboptimal strength, selectivity and size. Here, leveraging recent insights into the function of enhancers, we developed synthetic super-enhancers (SSEs) by assembling functionally validated enhancer fragments into multipart arrays. Focusing on the core SOX2-driven and SOX9-driven transcriptional regulatory network in glioblastoma stem cells (GSCs)1, we engineered SSEs with robust activity and high selectivity. Single-cell profiling, biochemical analyses and genome-binding data indicated that SSEs integrate neurodevelopmental and signalling-state transcription factors to trigger the formation of large multimeric complexes of transcription factors. Moreover, GSC-selective expression of a combination of cytotoxic (HSV-TK and ganciclovir) and immunomodulatory (IL-12) payloads, delivered using adeno-associated virus vectors, as a single treatment led to curative outcomes in a mouse model of aggressive glioblastoma. Notably, IL-12 induced an immunological memory that prevented tumour recurrence. The activity and selectivity of the adeno-associated virus and SSE were validated using primary human glioblastoma tissue and normal cortex samples. In summary, SSEs harness the unique core transcriptional programs that define the GSC phenotype and enable precision immune activation. This approach may have broader applications in other contexts when precise control of transgene expression in specific cell states is necessary. Synthetic super-enhancers enable specific delivery of anticancer payloads, achieving tumour elimination after a single dose in a mouse model of aggressive glioblastoma. Destination vector cloning We built a specific custom destination vector for efficient Golden Gate cloning of enhancer fragments. This included the reporter gene cassette NanoLuc-Ires-mNGreen-pA, downstream of a mCMV promoter. A bacterial suicide ccd... [48211 chars]