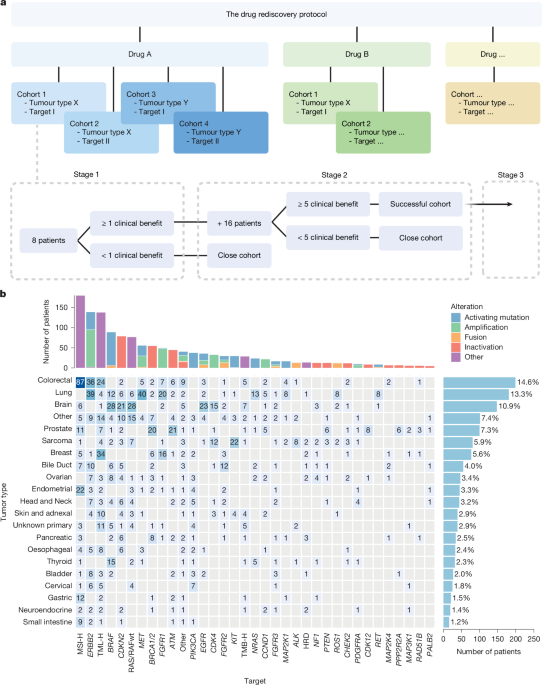
Anticancer drugs are frequently used off-label for tumours that are genetically similar to the approved indication. However, outcomes are rarely captured systematically, limiting evidence-based decision-making and risking repeated futile treatment. The Drug Rediscovery Protocol (DRUP; ClinicalTrials.gov ID: NCT02925234 ) prospectively evaluates such off-label use in patients in the Netherlands with advanced solid tumours who lack standard treatment options and harbour actionable genomic alterations1. Here we present results of 1,610 patients who began treatment with 37 different off-label drugs between July 2016 and May 2024 in the DRUP trial. Of these patients, 1,363 were response-evaluable, including 533 (39.1%) with rare cancers. The clinical benefit rate (confirmed response or stable disease for at least 16 weeks) was 34.9% (95% confidence interval, 32.2–37.6) and the objective response rate was 15.7% (95% confidence interval, 13.7–17.9). Median progression-free and overall survival were 3.4 months (95% confidence interval, 2.8–3.5) and 8.2 months (95% confidence interval, 7.6–8.8), respectively. Grade 3 or higher treatment-related adverse events occurred in 28.4% of patients. Notably, evidence generated in DRUP was used for reimbursement decisions by the regulatory bodies in the Netherlands2. Although activity across all tumour–drug combinations was modest, defined molecular subgroups and exceptional responders (7.0%) achieved meaningful benefit. To maximize patient benefit, we recommend that off-label precision medicines should be used only within frameworks that systematically evaluate efficacy and toxicity, support biomarker refinement and enable stepwise assessment toward potential future label expansion. These frameworks should prioritize high-confidence targets, early intervention, regulatory-aligned end-points and international collaboration. Evaluation by the Drug Rediscovery Protocol of off-label use of 37 approved cancer drugs on 1,610 patients showed modest activities overall, although subgroups of responders suggest that off-label drug use could be beneficial in tandem with more stringent biomarker selection. Patients with advanced or metastatic cancer who have exhausted all approved treatments often seek additional therapeutic options. The rise of social media, patient advocacy groups and other information sources have made patients and physicians increa... [27286 chars]