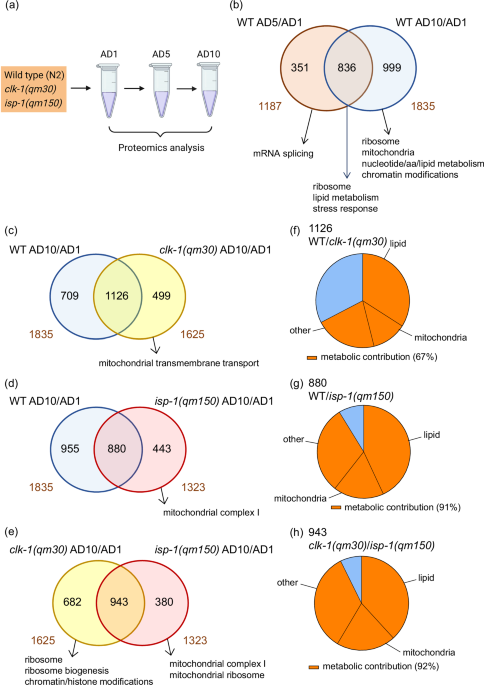
Mitochondrial dysfunction is a prominent hallmark of aging contributing to the decline of metabolic plasticity in late life. While genetic distortions of mitochondrial integrity elicit premature aging, the mechanisms leading to “natural” aging of mitochondria are less clear. Here we use proteomics, lipidomics, genetics and functional tests in wild type Caenorhabditis elegans and long-lived clk-1(qm30) and isp-1(qm150) mitochondrial mutants to identify molecular pathways that support longevity amid persistent mitochondrial inefficiency. These tests and subsequent transcriptomics and metabolomics analyses in humans reveal aging-associated decline of phosphatidylcholine synthesis as a trigger of mitochondrial network disruption, which contributes to mitochondrial dysfunction during normal aging. Moreover, ectopic boosting of phosphatidylcholine levels via diet restores late life mitochondrial integrity in vivo in nematodes and reinstates metabolic resilience in human cell culture tests. We thus describe a previously unrecognized natural driver of mitochondrial decline in aging that is malleable by dietary interventions. Mitochondrial decline impairs late-life metabolic plasticity. Using nematodes and human data, this study identifies reduced phosphatidylcholine synthesis as a natural trigger of mitochondrial dysfunction during aging. Proteomics analysis of wild type C. elegans and long-lived mitochondrial mutants reveals metabolic decline as a late event during aging In our search for malleable processes that can alleviate mitochondrial deterioration with age, we decided to analy... [60077 chars]